Summary
Cladiella pachyclados is an octocoral species that belongs to the Order Alcyonacea. The colony of C. pachyclados is sessile, firm and fleshy and has a stubby lobed form which gives it a very lumpy appearance.Colonies are generally gray-white or pale brown in colour contrast to the brown polyps. A significant characteristic of this species is the quick retraction of polyps which causes an instantaneous change from brown to white of the colony.
C. pachyclados is commonly found attached on hard substrates on rocky coasts with high wave intensity in order to eliminate the mucus produced by the colony. The presence of zooxanthellae in the soft coral allows it to use photosynthesis to meet its energy requirements. Besides that, the colonies also capture tiny food particles and absorb the dissolved organic matter. This species reproduce sexually through broadcast spawning. The polyps possess eight fringed tentacles which is a common characteristic of octocorals. Sclerites of the C. pachyclados are tiny and generally roughended dumbbell shapes. The Cladiella species is widely distributed and mainly spotted in the Red Sea and Indian Oceans. This species is not listed on the IUCN Red List for Endangered Species.
This webpage hopes to provide more detailed information on this Cladiella species as literature found is very limited. Three different experiments were conducted in the aspects of Chemical Ecology, Reproduction and Propagation and Sclerites Observation.
Physical Description
The colonies of Cladiella pachyclados are like many other soft coral, they are soft and commonly firm, fleshy and several stout lobed form which gives a lumpy appearance. The colonies are relatively insignificant and have a restricted polyp contraction control (Fabricius & Alderslade 2001). They possess a small but very distinctive stalk at the base that attaches the coral to rocks. The polyparium of the colony, which is the supporting framework of the coral, are divided into many short, rounded knob-like bump (Fabricius & Alderslade 2001). The stalk of this coral is generally white, grey or pale brown in color when the polyps are contracted.

However when the polyps are expanded, the colony will be brown or greenish due to the dense concentration of zooxanthellae in the polyps.

The quick retraction of polyps when disturbance occurs causes an instantaneous color change from brown to white. This is one of the interesting characteristic of the coral which also gives rise to its common name, the blushing coral (Fabricius & Alderslade 2001).
Ecology
Most soft corals are susceptible to abrasion and damages done by extreme tidal waves however, these thick and encrusting soft coral species are able to withstand the wave action. Hence, they can be found normally in the coastal waters on crests or rocky shores in wave-exposed environments (Fabricius & Alderslade 2001). They are scarce and rarely be found in a broad range of other reef habitats as the colonies of Cladiella pachyclados tend to secrete mucus therefore requires strong wave action on a daily basis to remove the mucus from the surrounding of the coral (Thiel 1997).
The colonies of Cladiella pachyclados are found attached to boulders or hard substrates on the south side outer-shelf of the Heron Island Reef. The physical environment found here is mainly high visibility water with low particle loads and slight resuspension of sediments. Disturbances by terrestrial run-off are absent however occasional upwelling of nutrient-enriched deeper water occurs here.
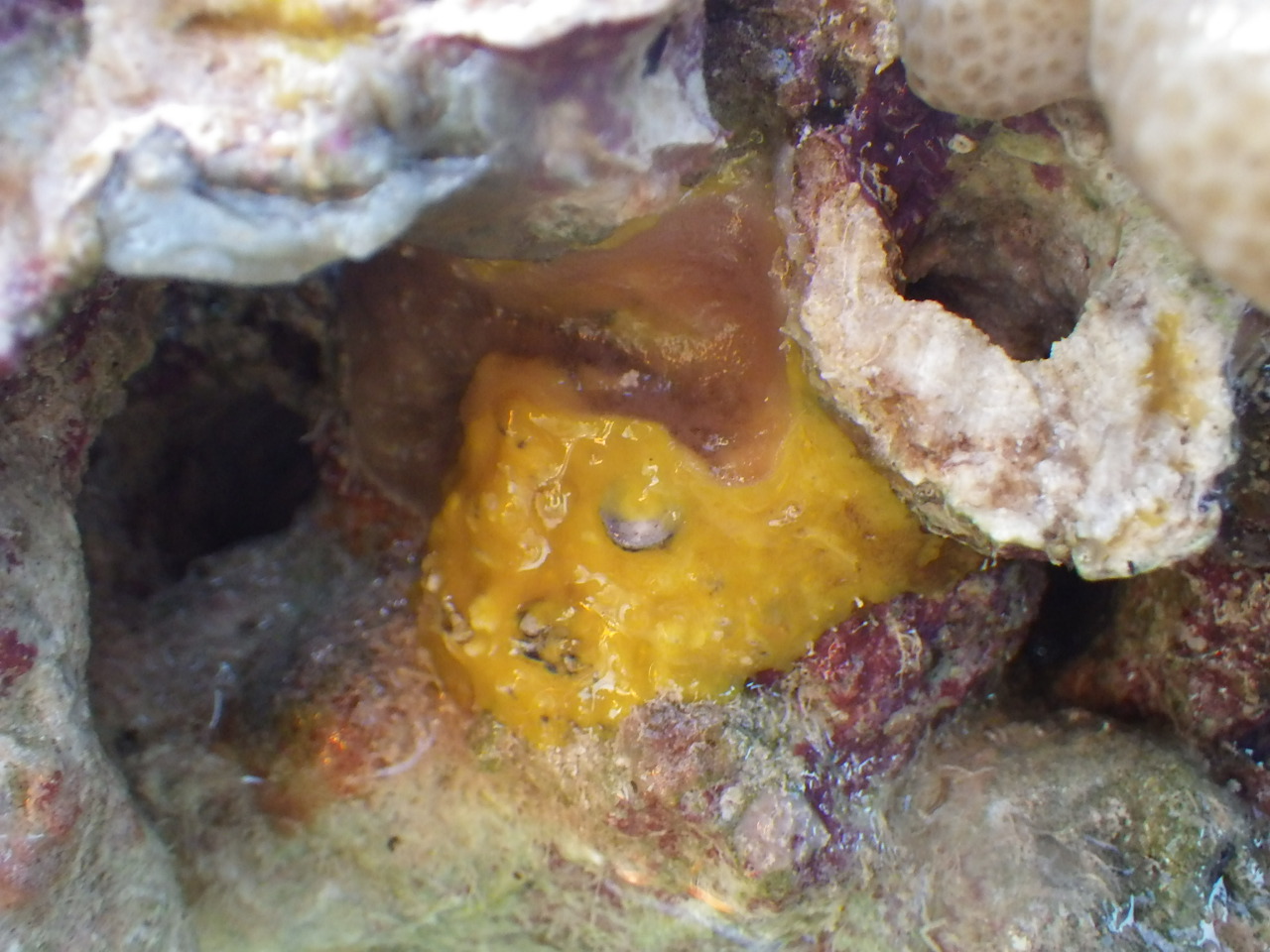
The colony tends to attach onto boulders and rocky substrates. It was discovered that there is a variety of invertebrate community living together with the coral. For example, the holothurian (sea cucumber) shown in the picture below. It is hypothesized that this sea cucumber uses the similarity in colour to the coral to camouflage itself by other predators as it burrows among the boulder and coral. The second picture shows a crab living within the damaged tissue of the colony. The last picture shows a sponge living on the boulder.


Chemical Ecology
Soft corals are able to produce a rich source of chemical substances as they are mostly sessile during the adult phase. Their inability to escape from predators, competitors fighting for space and ultraviolet radiation exposure causes them to develop these bioactive molecules in order to discourage predators, prevent competitors to overgrow them and filter from harmful radiation (Fabricius & Alderslade 2001). The group of chemicals identified as terpenoids are toxic and capable to reduce palatability are responsible of deterring predators from feeding on the corals (Fabricius & Alderslade 2001). Besides that, the soft corals are also capable in producing anti-fouling substances which prevent overgrowth of algae or fungi on the colonies. In addition to that, soft corals such as Sinularia and Sarcophyton species are able to release allelopathic substances into the water to hinder neighbouring animals or plants to overgrow and compete for space (Fabricius & Alderslade 2001). Shallow water organisms including these soft corals have developed unique compounds known as mycosporine-like amino acids (MAAs) to screen out the high ultraviolet radiation exposure (Fabricius & Alderslade 2001).
Studies were done to determine the chemical constituents of the genus Cladiella soft corals by various research groups as this genus is proved to have a rich supply of cytotoxic eunicellin-based sesquiterpenes and diterpenes. Cladiellin, acetoxycladiellin, cladioxazole and cladidiol are some of the examples that belong in this group (Radhika 2006). The most significant breakthrough related to the Cladiella pachyclados is the discovery of 5 new eunicellin diterpenes which are the pachycladins A-E. These diterpenes were found to show significant anti-invasive and migratory inhibitory activities of the prostate cancer cells (Hassan et al. 2010).
The next group is the ceramides, cerebrosides, lipids and glycolipids (Radhika 2006). A combination of cerebroside homologues and a lipid glycoside was researched independently and found that they have a significant antifungal and antibacterial activity (Radhika 2006). The last group of chemical constituents discovered are the sterols and sterol glycosides (Radhika 2006). In conclusion, 55 complementary metabolites have been isolated from the various Cladiella species and further studied by researchers.
Project 1:
The effect of water flow on the production and accumulation of mucus in the surrounding of Cladiella pachyclados.
Hypothesis:
Mucus produced by the colony tends to aggregates around it and potentially affects the coral health under low water flow.
Methods:
1. Two colonies were obtained and placed in separate aquariums.
2. One of the colonies was placed under strong to moderate water flow and the other in slow to no water flow.
3. Observations were made over time.
Results:
As shown in the picture below, mucus coatings are seen in the slow to no water flow aquariums whereas in the strong to moderate water flow, no mucus aggregation is seen.

Discussion:
Some soft coral species are known to occasionally excrete a film of waxy mucus when stressed. The mucus functions as a chemical shield or to clean the colony surface from sediments and fouling organisms (Fabricius & Alderslade 2001). Accumulation of these mucus coatings leads to a build-up of bacteria and thus causes illness to the colonies. Therefore, moderate to high water flow is adamant for the Cladiella pachyclados to eliminate the mucus from the surroundings of the coral (Thiel 1997). This explains the need for the colonies to reside in the coastal waters on crests or rocky shores with high wave action in order for the current to remove the mucus and leave a clean colony. However, due to insufficient time on the Heron Island, this experiment was unable to be carried out to investigate the time taken and the quantity of mucus produced by the coral which will cause serious illness to the colonies.
Life History & Behaviour
Feeding:
The Cladiella pachyclados contains symbiotic algae zooxanthellae in its tissue which are photosynthetic. These zooxanthellae use sunlight, water and carbon dioxide to produce sugars and other cellular materials which are then transfer to the coral (Fabricius & Alderslade 2001). In return, the coral provides a protected microhabitat for the algae to live in and unlimited nutrients and carbon dioxide resources. Therefore, high but not too excessive light intensity environment is very much needed in order for these coral colonies to grow. Besides that, the polyps that emerge from the branches of the colonies can trap tiny suspended food particles to feed on (McBirney & Brough nd). Flow rate of the water and food concentration is essential in affecting the nutrients intake and development in soft corals.
Movement:
C. pachyclados is a sessile species as it mainly attaches to rocks, boulders or any hard substrates and propagates.
Growth Rate:
According to studies done by Fabricius (1995), C. pachyclados found on the reef flats of Heron Island demonstrates a slow growth rate compared to other opportunistic and fast growing soft corals such as the Alcyonium siderium.
Reproduction and Propagation
The reproductive biology of most soft coral species are poorly known as studies are less conducted on soft corals compared to the hard corals. The current knowledge of soft coral reproduction is limited and only includes the reproductive morphology on mostly tropical species (Simpson, nd). The soft corals exhibit both asexual and sexual reproduction. However, current studies show that asexual reproduction is more common in soft corals. Various types of asexual reproduction occur in the soft corals which include simple fission, reattachment of colony fragments and parthenogenesis (Benayahu and Loya 1985; Walker and Bull 1983; Hartnoll, 1975). The population growth rate of these asexual reproduced soft corals is revealed to be greater as colonization is more rapid and thus will allow a quick recover from disturbance (Benayahu and Loya, 1987).
Sexual reproduction in soft corals occurs in two ways, broadcast spawning which fertilization and development occurs in the water column or fertilization and brooding of embryos takes place internally or externally on the maternal colony (Simpson, nd). Majority of the soft coral species that reproduce sexually are gonochoristic which means separate sexes, however hermaphroditism (both sex in an individual) was also recorded in some soft corals. According to Coll et al. (1995), broadcast spawning is the most common strategy used by the Alcyoneans to sexually reproduce whereas brooding occurs mostly in the gorgonian type of corals. The broadcast spawning soft coral colonies tend to partake in synchronized mass spawning events to decrease predation pressure and in turn increase reproductive success rate (Alino and Coll, 1989).
Project 2:
Forced spawning of eggs in Cladiella pachyclados and the observation of eggs extracted.
Hypothesis: Desiccation of the colony and insertion of potassium chloride into the environment may force the coral to spawn and the eggs that will be excreted will be observed.
Methods:
1. Two colonies are placed in separate aquariums with one aquarium filled with sea water until the coral is submerged in it and the other is empty.
2. Approximately 4g of potassium chloride is added into the filled sea water aquarium and left in a room temperature area.
3. The other colony that is placed in the empty aquarium is left under the sun for approximately 2 hours.
4. The desiccated individual is then brought in and submerged in sea water for 2 hours.
5. Observations are made and evaluated.
Results:
Slight spawning is observed in the first aquarium which the colony is immersed in sea water with potassium chloride. However, no spawning has occurred in the desiccated treatment. The eggs excreted in the first treatment appear to be oval shaped and white in colour (shown in the picture below).

Discussion:
According to studies done by Horridge (1956), potassium ions act as a chemical stimulation to the soft corals. It triggers a slow response of the polyps and causes the colony to spasm and eventually relaxes and paralyze due to the hyperactivity of the cilia. Although under a calm and insensitive state, the muscles continue to contract and push the eggs out through the wide open polyps and thus force spawning occurs. Only eggs were found to be excreted but no sperm was released. This can be explained by the studies which demonstrate that C. pachyclados undergoes a broadcast spawning reproductive strategy and has separate male and female reproductive structures (Simpson, nd). Therefore, it is unable to show any fertilization and larval development occasion. This may act as a stepping stone for further research to be held on the reproductive biology of C. pachyclados. The desiccation treatment is not successful in forcing the colony to spawn can be explain by the insufficient time for the colony to be paralyze to trigger spawning.
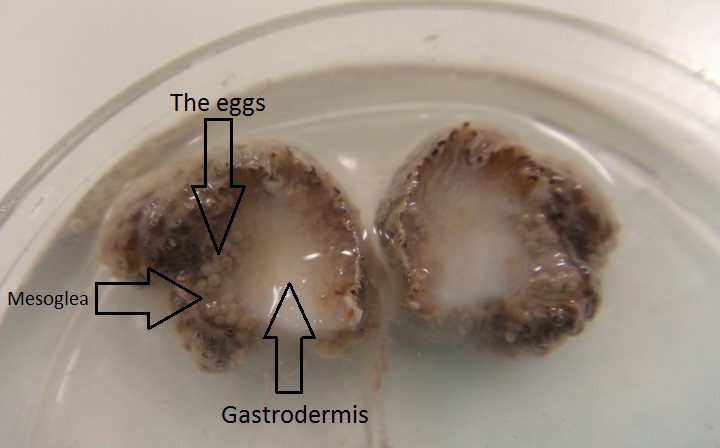
Due to the evidence that eggs are found in the experiment, few small projections of different individual were dissected through the middle to examine the position of the eggs. As seen in the picture below, the eggs appear to be white and big in size. They are mostly embedded in the mesoglea and a greater quantity is found to be concentrated at the base compare to other parts of the projection. When the projection is sliced through, eggs that are readily released into the environment are hypothesized to be mature eggs. Whereas the immature eggs mostly stay in sacs of few and firmly embedded in the mesoglea are not easily acquired.
Anatomy & Physiology
Polyp anatomy
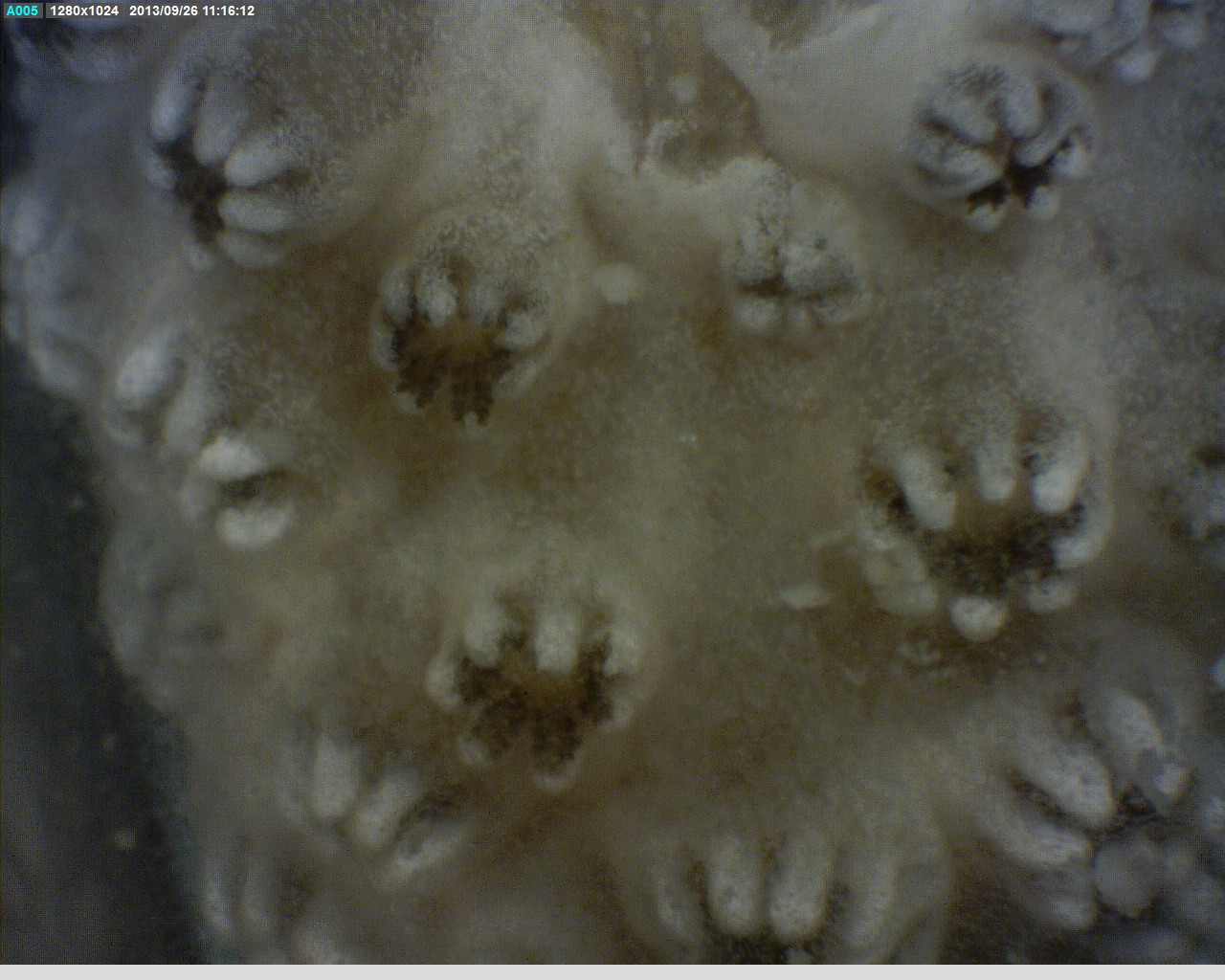
Picture below shows a close up picture of a branch of the colony with open polyps.

Majority of soft corals have the autozooid type of polyp (Fabricius & Alderslade 2001). It is responsible for capturing food and reproduction. The other type of polyp is the siphonozooid which functions to irrigate colonies with seawater and transport dissolved and tiny food particles suspended in the water column (Fabricius & Alderslade 2001). The soft coral polyp varies from the hard corals by the absence of solid limestone skeleton which is replaced by limestone crystal structures known as sclerites that are embedded in the tissues (Fabricius & Alderslade 2001). Besides that, the structure of the soft coral polyp is similar to hard corals, however they have eight fringed tentacles which identifies them as a soft coral (shown in the picture below). The polyps of C. pachyclados are retractile and minute. They have very short bodies and are monomorphic. The tentacle tips tend to bend backwards to create an inverse bowl shape when expanded (Fabricius & Alderslade 2001).
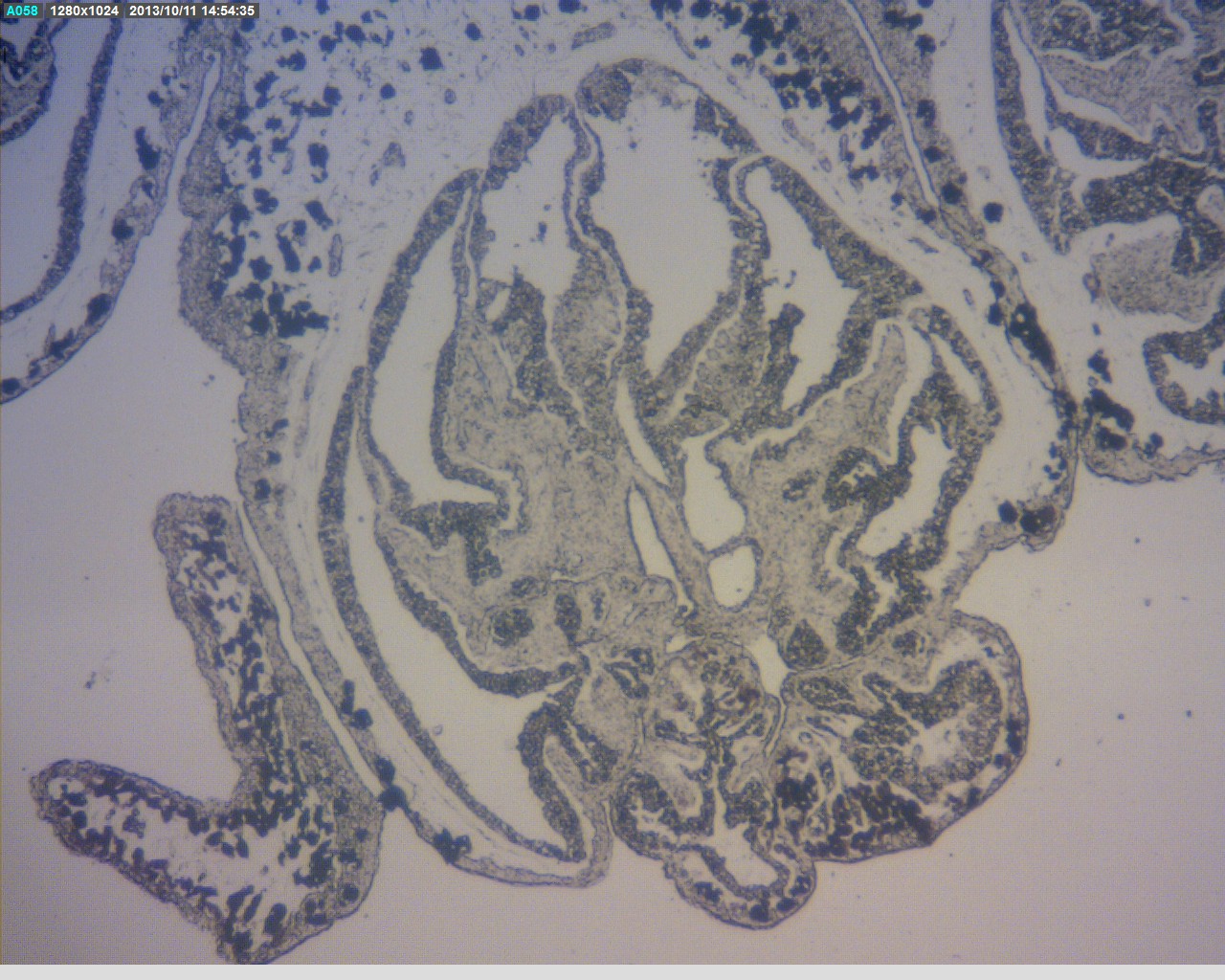
The picture below shows a cross section of the C. pachyclados polyp. The little black dots signify zooxanthellae embedded in the polyp. This picture also clearly shows the presence of eight fringed tentacles in C. pachyclados.
A closer look at the dense concentration of zooxanthellae embedded in the polyps. The presence of zooxanthellae gives rise to the brownish colour of the polyps.
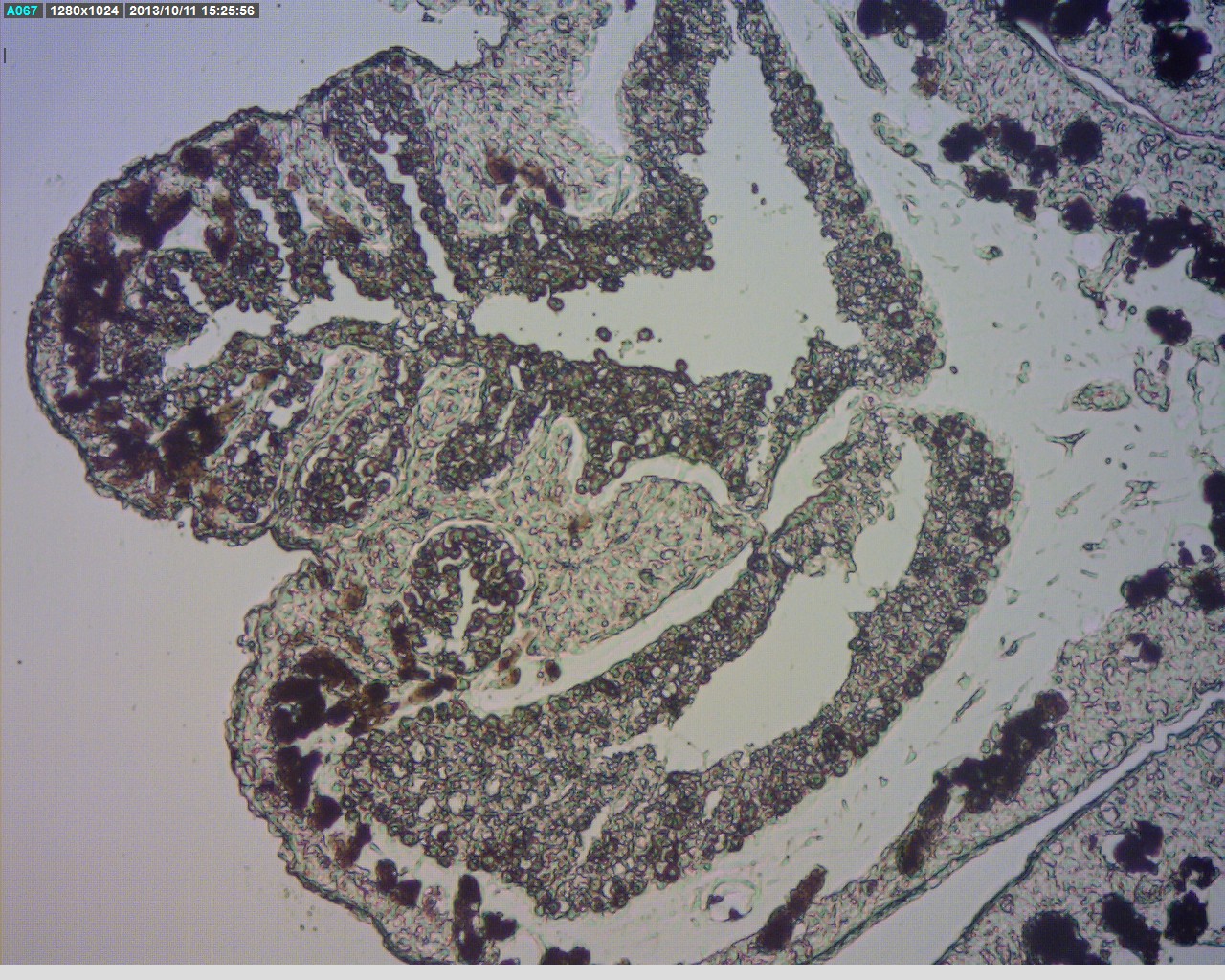
One of the interesting characteristics of the C. pachyclados is the ability of the polyps to close up instantaneously upon disturbance. However, as discussed in the forced spawning experiment, the incorporation of potassium chloride into the environment of the colony has disabled the polyps to close immediately as opposed to those without the presence of potassium chloride. As demonstrated by Horridge (1956), excess potassium ions in the living environment of the colony are capable to slow the tentacles responses and decrease the sensitivity of the colony to any contact. The video below shows the response of polyps to excess introduction of potassium ions into the environment as opposed to the colony without potassium incorporation respectively.
Sclerites
Some colonies of the Cladiella are very similar in shapes and sizes to the Alcyonium and Sinularia (Fabricius & Alderslade 2001). Therefore, in order to positively confirm the genus of these corals, an examination of the sclerites was carried out.
Project 3:
Sclerites observation to clearly identify the genus of the selected soft coral.
Hypothesis: The sclerites should be dumbbell shaped or roughended double-heads as the soft coral was hypothesized to be Cladiella species.
Methods:
- A small fraction of the coral is acquired and placed in a plate.
- Few drops of bleach is added into the fraction and left for 5 minutes.
- The fraction is then grinded into little pieces and the smashed pieces are placed onto a microscope slide using a pipette.
- Observations are made using the microscope.
Results:
As shown in the picture below, the sclerites found under the microscope are very minute and roughended dumbbell shaped. The quantity of sclerites discovered is not very significant.

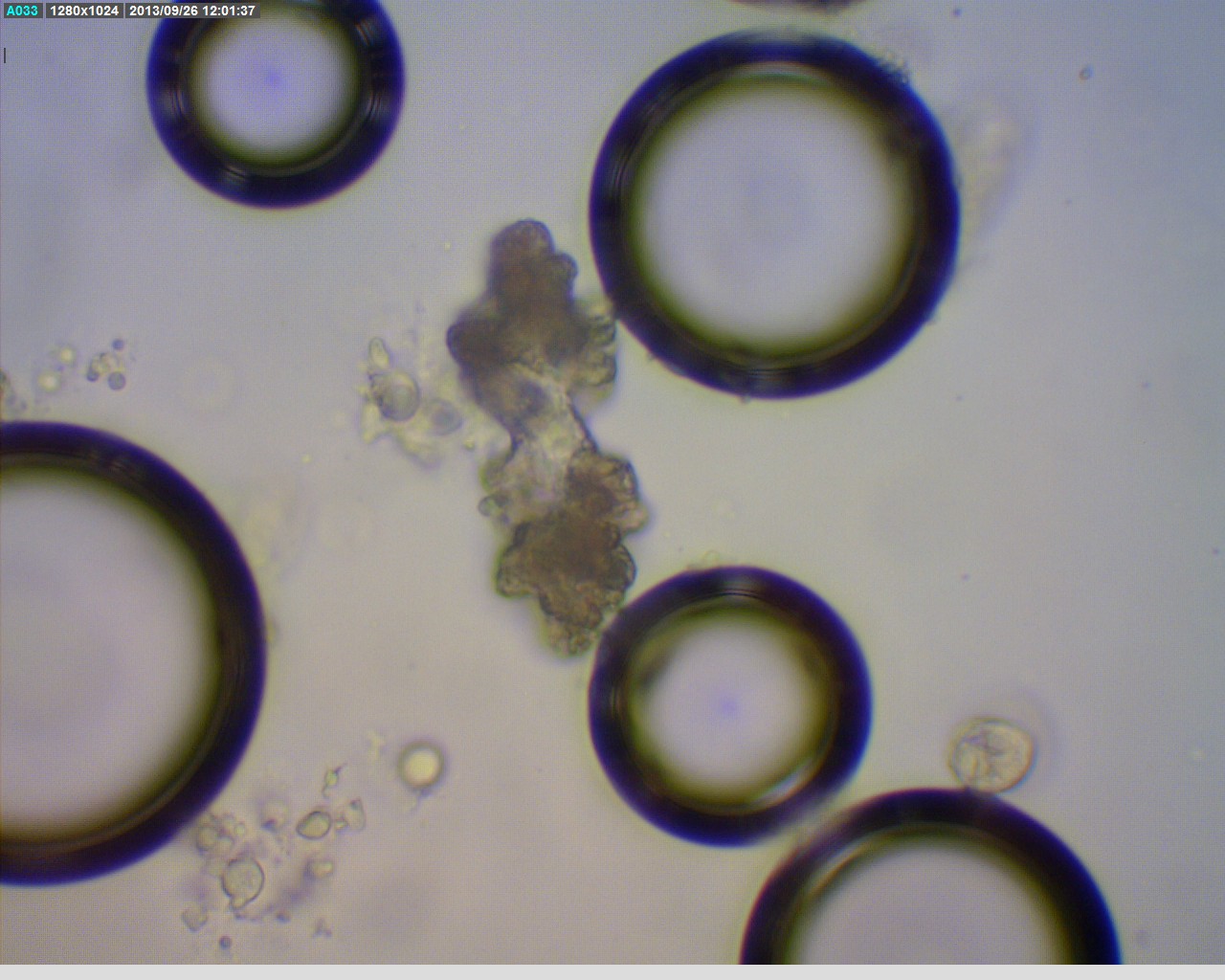
Discussions:
The discovery of great amount of roughended dumbbell shaped sclerites reveal that the soft coral selected is indeed a Cladiella species (Fabricius& Alderslade 2001). The insignificant quantity suggests that further grinding of the fraction may be required in order to extract more sclerites.
Evolution & Systematics
The current knowledge concerning the octocoral systematic is very ambiguous and inadequate due to the lack of studies conducted on these corals. Present literature is often confusing and variable as there is insufficient understanding on the soft coral species. Descriptions of these corals in the species level are also lack in detail and illustrations. The lowest level of identification of these soft corals only reaches the genus level.
Phylum Cnidaria:
Cnidarians are tentacle-bearing invertebrates that are usually radially symmetrical. They possess stinging structures known as Cnidocytes which is a distinguishing characteristic of this phylum. The cnidarians exist in two main growth stages which are polyp (solitary) and medusa (free swimming) forms. Both forms comprise the outer layer which is the epidermis that bears sensory cells and the inner layer, the gastrodermis that is responsible for digestion and reproduction. Mesoglea, a third layer connects both the outer and inner layer.
This phylum is split up into different classes that include the:
- Class Hydrozoa: Hydroids and stylasterine corals. This class is represented by both polyp-bearing and medusoid forms.
- Class Scyphozoa: The true jellyfish. This class mainly exists in free-swimming medusoid forms. However during their development, they are found attached by a stalk.
- Class Cubozoa: Box jellyfish. This marine jellyfish has a cubic bell shape hence the name of this class.
- Class Anthozoa: Soft and hard corals, sea pens, anemones and corallimorpharians. This class does not have the medusa stage and the polyps are mostly solitary or colonial.
Class Anthozoa:
This class is separated into different subclasses such as the:
- Subclass Hexacorallia: Sea anemones, hard corals and zoanthids. The polyps bear six or multiples of six tentacles without pinnules.
- Subclass Octocorallia:Soft corals, sea fans and sea pens. Each of the polyps comprise of eight hollow tentacles.
Subclass Octocorallia:
The subclass octocorallia is differentiated into different orders which are the:
- Order Helioporacea: Blue coral. The only octocoral that forms a massive aragonite skeleton like the hard corals.
- Order Pennatulacea: Sea pens. This group has a great diversity but lack in knowledge. Many species can be found in shallow water regions however majority lives in deep sea. They are usually characterised by the large, central axial polyp, known as the oozoid. This forms the penducle or foot of the colony and the autozooid and siphonozooid polyps.
- OrderAlcyonacea: This group is usually found in shallow waters of the Indian Ocean and Red Sea.
Family Alcyoniidae:
The colonies in this family are represented by the dominant reef-dwelling soft coral. Most colonies have a bare basal part and divided into lobed branches that bear the polyps. Some colonies have a flat upper surface.
Genus Cladiella:
Some colonies are mistaken as Sinularia or Klyxum. However, according to literature the lobes of Cladiella are shorter and more densely packed. The surface of the base does not feel rough as the sclerites are too minute. Nevertheless, in order to clearly identify the colonies, an examination on the sclerites can be carried out.
*The above information is retrieved from Ruppert, E Fox, R Barnes, R 2004, Invertebrate Zoology, 7th edn, Brooks/Cole Cengage Learning, Belmont, USA and Fabricius, K & Alderslade, P 2001, Soft Corals and Sea Fans: A comprehensive guide to the tropical shallow-water genera of the Central-West Pacific, the Indian Ocean and the Red Sea, Australian Institute of Marine Science, Queensland, Australia.
Biogeographic Distribution
This species is widely distributed and can be found in the Red Sea, Western Pacific to Polynesia and Indian Oceans.
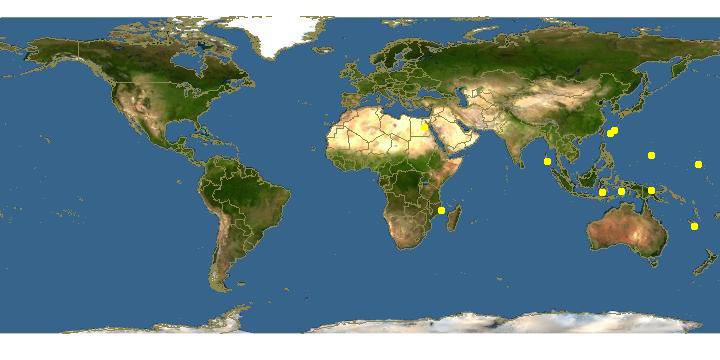
Image obtained from Discover Life. org (http://www.discoverlife.org/mp/20q?search=Cladiella+pachyclados)
Conservation & Threats
The threat status of Cladiella sp. is not evaluated by the International Union for Conservation of Nature (IUCN). Hence, it is not listed on the IUCN Red List for Endangered Species.
References & Links
Alino, PM & Coll, JC 1989, ‘Observations of the synchronized mass spawning and post settlement activity of octocorals on the Great Barrier Reef, Australia: Biological Aspects’, Bulletin of Marine Science, vol. 45, no. 3, pp. 697-707.
Benayahu, Y & Loya, Y 1985, ‘Settlement and recruitment of a soft coral: Why is Xenia macrospiculata a successful colonizer?’, Bulletin of Marine Science, vol. 36, no. 1, pp. 177-188.
Benayahu, Y & Loya, Y 1987, ‘Long-term recruitment of soft-corals (Octocorallia: Alcyonacea) on artificial substrata at Eliat (Red Sea)’, Marine Ecology Progress Series, vol. 38, pp. 161-167.
Coll, JC Leone, PA Bowden, BF Carroll, AR Koenig, GM Heaton, A Nys, R Maida, M & Alino, PM 1995, ‘Chemical aspects of mass spawning in corals. 2. (-)-Epi-thunbergol, the sperm attractant in the eggs of the soft coral Lobophytum crassum (Cnidaria: Octocorallia)’, Marine Biology, vol. 123, pp. 137-143.
Fabricius, KE 1995, ‘Slow population turnover in the soft coral genera Sinularia and Sarcophyton on mid- and outer- shelf reefs of the Great Barrier Reef’, Marine Ecology Progress Series, vol. 126, pp. 145-152.
Fabricius, K & Alderslade, P 2001, Soft Corals and Sea Fans: A comprehensive guide to the tropical shallow-water genera of the Central-West Pacific, the Indian Ocean and the Red Sea, Australian Institute of Marine Science, Queensland, Australia.
Hartnoll, RG 1975, ‘The annual cycle of Alcyonium digitatum’, Estuarine, Coastal and Shelf Science, vol. 3, pp. 71-78.
Hassan, HM Khanfar, MA Elnagar, AY Mohammed, R Shaala, LA Youssef, DTA Hifnawy, MS & El Sayed, KA 2010, ‘Pachycladins A-E, Prostate Cancer Invasion and Migration Inhibitory Eunicellin-Based Diterpenoids from the Red Sea Soft Coral Cladiella pachyclados’, Journal of Natural Products, vol. 73, no. 5, pp. 848-853.
Horridge, GA 1956, ‘The Responses of Heteroxenia (Alcyonaria) to Stimulation and to Some Inorganic Ions’, The Journal of Experimental Biology, vol. 33, pp. 604-614.
McBirney, C & Brough, C nd, Colt Coral, Animal-World: Pet and Animal Information, viewed 15 October 2013, <http://animal-world.com/Aquarium-Coral-Reefs/Colt-Coral#Status>.
Radhika, P 2006, ‘Chemical constituents and biological activities of the soft corals of genus Cladiella: Areview’, Biochemical Systematics and Ecology, vol. 34, no. 11, pp. 781-789.
Ruppert, E Fox, R Barnes, R 2004, Invertebrate Zoology, 7thedn, Brooks/Cole Cengage Learning, Belmont, USA.
Simpson, A nd, Reproduction in Octocorals (Subclass Octocorallia): A Review of Published Literature, University of Maine, Darling Marine Center, viewed 17 October 2013, <http://www.ucs.louisiana.edu/~scf4101/Bambooweb/repro_AS.html>.
Thiel, AJ 1997, Cladiella Corals –The Colt Corals, 3 February, viewed 21 October 2013, <http://netclub.athiel.com/cladiella/cladiella.html>.
Walker, TA & Bull, GD 1983, ‘A newly discovered method of reproduction in gorgonian coral’, Marine Ecology Progress Series, vol. 12, pp. 137-143.
|